 In the water the flow is irregular and chaotic, otherwise known as turbulent flow. In water the same balls will sink with non-uniform motion. In glycerin the balls should fall with a constant velocity. For the purposes of our demonstration we will keep both tubes at room temperature(20° C) with little to no fluctuation in heat. For liquids, when temperature increase the viscosity decreases. For gases, when temperature increases the viscosity increases. Viscosity is also very temperature dependent. The pascal second is the SI unit for viscosity. It is given as the velocity of the moving surface divided by l (the original length of the material). Strain rate is the rate of change of deformation or strain in a material. It is defined as the force tangent to the material surface divided by the area in which it acts. Shear stress is the the friction between fluid particles sliding past each other. Viscosity is denoted by η (eta) as the ratio of the shear stress (Force/Area) to the strain rate. Viscous forces oppose the motion of one portion of the fluid relative to another. Viscosity is internal friction inside a fluid. Is it close to the value you found experimentally? Alternatively use the viscosity of glycerin to calculate the terminal velocity. 
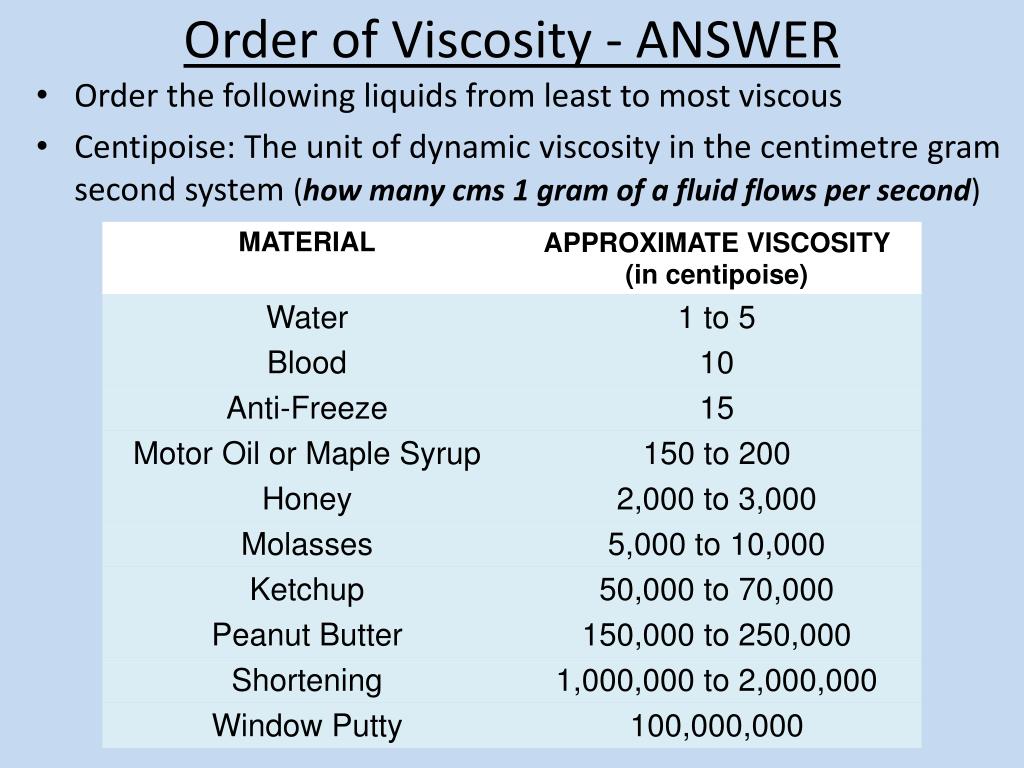
Also, compare it to the given values in the table below.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
